Organoids in cancer research: an interview with Luke Boulter
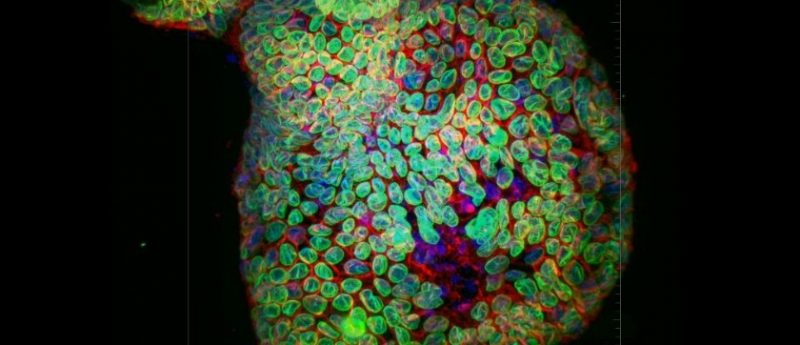
Photo credit: Mouse Colonic Epithelial Organoid. Confocal image captured with iXon Ultra 888- 20x water immersion with 40um pinhole (Andor Technology). Ronan Mellin, Dr. Luke Boulter, MRC Human Genetics Unit
We recently had the chance to speak with Luke Boulter (University of Edinburgh, UK) about his research into how signals from the regenerative microenvironment drive tissue repair following injury and how these signals are co-opted by the tumor to support cancer growth. In their research, Boulter’s lab utilizes organoids to study liver tissue; find out about the potential of organoids in cancer research, including techniques to generate them and their key advantages and challenges in this interview.
Could you briefly introduce yourself and tell us more about your research?
I’m a basic scientist who is essentially interested in how cells form ducts and tubes. In the developing liver, bile ducts, tubes that carry bile into the bowel need to form, but it’s not particularly clear how this happens. Importantly (and unusually), the adult liver is really good at repairing itself, but that presents a duct with a new set of challenges – how do you make a duct when you need to maintain the function of a tissue? If ducts continue to grow then they can form bile duct cancers, which are particularly aggressive – only 1-in-20 people diagnosed with cholangiocarcinoma will survive more than 5 years, so we urgently need to understand how these cancers grow to develop new therapeutic approaches.
…we can expand the number of organoids by taking them out of these 3D gels and then breaking them into small fragments each of which form an organoid of their own…
How do you generate organoids from human bile ducts/ cholangiocarcinoma?
We start by taking a piece of liver tissue (normally from a liver that has been rejected for transplant) or from a resected cancer. These pieces of tissue are digested and then ducts and cancer cells that are isolated from these are grown in a 3D extracellular matrix, which when it gels allows organoids to grow – we can expand the number of organoids by taking them out of these 3D gels and then breaking them into small fragments each of which form an organoid of their own. The culture conditions for our organoids are quite specific and a lot of work has been done by other labs to develop culture conditions for organoids from a variety of tissues.
Are there particular cancer indications that are better suited to organoid research?
To my knowledge many of the organoid systems have been derived from very epithelial tumors. There are few systems in place to grow tumors with a mesenchymal phenotype – again, this limits our ability to use these systems to screen compounds in a number of cancer phenotypes.
In terms of organoid production and application in cancer research, where do you hope the field will be in the next 5 years?
I am sure we will see the expansion of organoid libraries and biobanks over the next few years, certainly for cancers that form organoid easily and that can be easily propagated – large libraries already exist for colorectal and ovarian cancers for example. I would really like to see organoids grown with more non-cancer cells in the future, to really reflect the tumor organ. We also need to address the elephant in the room that many of these organoid lines are grown in a basement membrane that doesn’t really reflect the composition of the tumor extra cellular matrix – making patient matched extra cellular matrix s I think would be a pipe-dream, but developing matrices that more closely reflect the cancer cell environment will be important for understanding how these organoids respond to therapeutics.
I am sure we will see the expansion of organoid libraries and biobanks over the next few years, certainly for cancers that form organoid easily…
What advice/tips would you give to other cancer researchers who are considering to use organoids?
Have a go and talk to people who have experience in growing cancer organoids. They are not trivial; however, they can also be an important tool for testing biological and pharmacological questions.
In your opinion, what are the main advantages of using organoids for research compared with 2D or other 3D cultures?
For us, the structure of a duct, be it a healthy one or a cancerous one is important – cells are polarized, they have a top and bottom face and they also have a lumen. 2D cell cultures just cannot mimic this level of architecture and so organoids are a great resource. We have also found that it is relatively straight forward to dissociate organoids into fragments that can be infected with lentiviruses – we can edit genes and over express proteins with relative ease making these structures preferable to other 3D cultures which can’t be manipulated so easily.
What are the key challenges you face when using organoids in your work?
I think there are two main challenges to our organoid work. The first is cost – the price differential between organoid cultures and 2D cultures is significant and it means that we as a research group are very conscious of how and when we use organoid cultures. Organoid cultures are also technically challenging, particularly from cancer tissues – we had a number of aborted attempts before we managed to grow these organoids and from our experience and from the experience of others not all cancers (even of the same type) can equally generate organoids.
Related content:
- Organoid study sheds light on how colorectal cancer cells may avoid immunotherapy
- What is the importance of organoids in personalized medicine?
- Organoids as a model system in oncology: an interview with David Tuveson

Profile: Luke Boulter is Chancellor’s Fellow and Principal Investigator at the MRC Institute of Genetics & Molecular Medicine, MRC Human Genetics Unit, at the University of Edinburgh (UK). Boulter moved to the MRC Human Genetics Unit in 2014 as a Leverhulme Trust Early Career Fellow and University of Edinburgh Chancellors Fellow, his lab’s work focuses on how Wnt signalling regulates the biology of the liver in health and disease. They use a combination of human genetics, molecular biology and disease modelling to ask fundemental questions about how Wnts function in vivo and try to apply this knowledge to developing therapies for liver disease.