Publication In Focus: TALAPRO-2
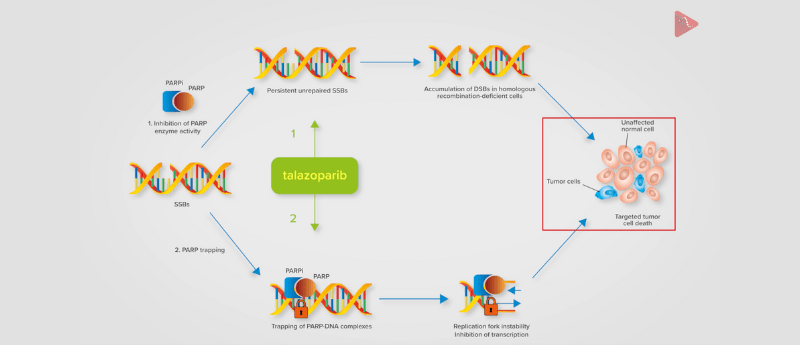

PARP inhibitors in combination with androgen receptor-targeted therapy have demonstrated potential in the treatment of metastatic castration-resistant prostate cancer (mCRPC). A recent clinical trial protocol, published in Future Oncology, described the design and rationale of the multinational, Phase III, two-part TALAPRO-2 study comparing talazoparib plus enzalutamide versus placebo plus enzalutamide as a first-line treatment for patients with mCRPC with or without DNA damage response alterations.
This Publication In Focus showcases content that provide further details of the trial in various multimedia content types. Hear Neeraj Agarwal (University of Utah, UT, USA) discuss the recent publication in the Video Journal of Biomedicine’s video or gain a visual snapshot of the Phase III trial by checking out the infographic below. Finally, the plain language summary can help patients, their family members and caregivers understand the article.
FEATURES
 TALAPRO-2 study design infographic
TALAPRO-2 study design infographic
This infographic summarizes the study design of the TALAPRO-2 trial, including objectives, rationale and eligibility criteria. View the full infographic here >>>.

Read the original journal article, which describes the design and rationale of the Phase III, two-part TALAPRO-2 study comparing talazoparib plus enzalutamide versus placebo plus enzalutamide as a first-line treatment for patients with mCRPC.

Plain language summary of the design of the TALAPRO-2 study
Read the plain language summary for the above journal article, which is specifically designed to be read by patient and non-specialist audiences to help them to understand the research presented.

Video: TALAPRO-2 – talazoparib plus enzalutamide in metastatic castration-resistant prostate cancer
In this video, Neeraj Agarwal (University of Utah, UT, USA) on behalf of his co-authors discusses their recent publication. View the full video here >>>.